Is CBD legal?
In many parts of the country, CBD exists in a legal gray zone.

Photo by Jens Kalaene/picture alliance via Getty Images
If you’re confused about whether the CBD products flooding your city or town are totally above board, join the club. CBD is at the center of a complicated legal morass that’s in constant flux. Here’s what we currently know — and what’s accurate — at the date of publication.
This story discusses substances that are legal in some places but not in others and is for informational purposes only and not for the purpose of providing legal advice. You shouldn’t do things that are illegal — this story does not endorse or encourage illegal drug use.
What federal law says about CBD
The federal government recognizes two forms of the plant Cannabis sativa: hemp and marijuana. Hemp is the least processed form of the cannabis plant and contains high levels of cannabidiol, or CBD — the compound in cannabis known for its medicinal properties — and traces of delta-9-tetrahydrocannabinol, or THC, the psychoactive component in cannabis that causes a “high.”
Marijuana, on the other hand, is increasingly being bred to contain higher levels of THC — sometimes upward of 30% — and lower levels of CBD — an average of less than 0.2%, according to research.
In 2018, the Agriculture Improvement Act of 2018 (aka the Farm Bill) legalized CBD that is derived from hemp and contains no more than 0.3% THC (by dry weight). CBD that comes from the marijuana plant remains illegal under the Controlled Substances Act since the Drug Enforcement Administration (DEA) classifies marijuana as a Schedule I drug. In DEA parlance, that means the administration believes it has “no currently accepted medical use and a high potential for abuse.”
The Farm Bill also created regulations for hemp farmers, which means, “that any cannabinoid—a set of chemical compounds found in the cannabis plant—that is derived from hemp will be legal, if and only if that hemp is produced in a manner consistent with the Farm Bill, associated federal regulations, association state regulations, and by a licensed grower,” according to the Brookings Institute, a non-profit public policy organization.
In other words, if a CBD product contains the legal amount of THC but wasn’t grown by a licensed producer according to federal regulations, it’s still illegal.
The day the Farm Bill was signed into law, the US Food and Drug Administration (FDA) released a statement clarifying that Congress had “explicitly preserved the agency’s current authority to regulate products containing cannabis or cannabis-derived compounds under the Federal Food, Drug, and Cosmetic Act (FD&C Act) and section 351 of the Public Health Service Act.”

Justin Aikin/Unsplash
Because CBD is also an approved prescription drug (Epidiolex), the FDA still considers CBD a drug ingredient, which means it cannot be marketed and sold as a dietary supplement with therapeutic properties (or even shipped across state lines) without first going through the FDA’s drug approval process — regardless of whether the products are derived from hemp.
So, what about all the CBD pills, oils and edibles currently on the market that claim to fight anxiety, decrease pain and curb the spread of cancer? That’s what the FDA is currently trying to figure out.
None of these products have been proven to be safe or effective. The FDA has the power to give CBD the green light and the agency is currently considering whether to do that. They held a public hearing on May 31, 2019, and opened a docket for public comment through July 16, 2019.
In the meantime, it’s unclear whether the FDA will crack down on violators. According to the FDA, the agency considers many factors when deciding whether or not to initiate an enforcement action, including agency resources and the threat to public health.
What state laws say about CBD
Although hemp-derived CBD is now federally legal, many states and cities are updating their laws and they’re not always in line with federal regulations.
For instance, Colorado recently made it legal to use all parts of the hemp plant as a food ingredient, whereas products with any amount of THC are still illegal in Texas.
The best way to determine if CBD products are legal in a specific state is to become familiar with local cannabis laws.
Organizations like the National Conference of State Legislatures, the Marijuana Policy Project and the National Organization for the Reform of Marijuana Lawsmaintain databases — and interactive data visualizations — like the one below that make it easy to see what’s going on in each state, but for the most up-to-date information, should check the state’s criminal code or agriculture department.
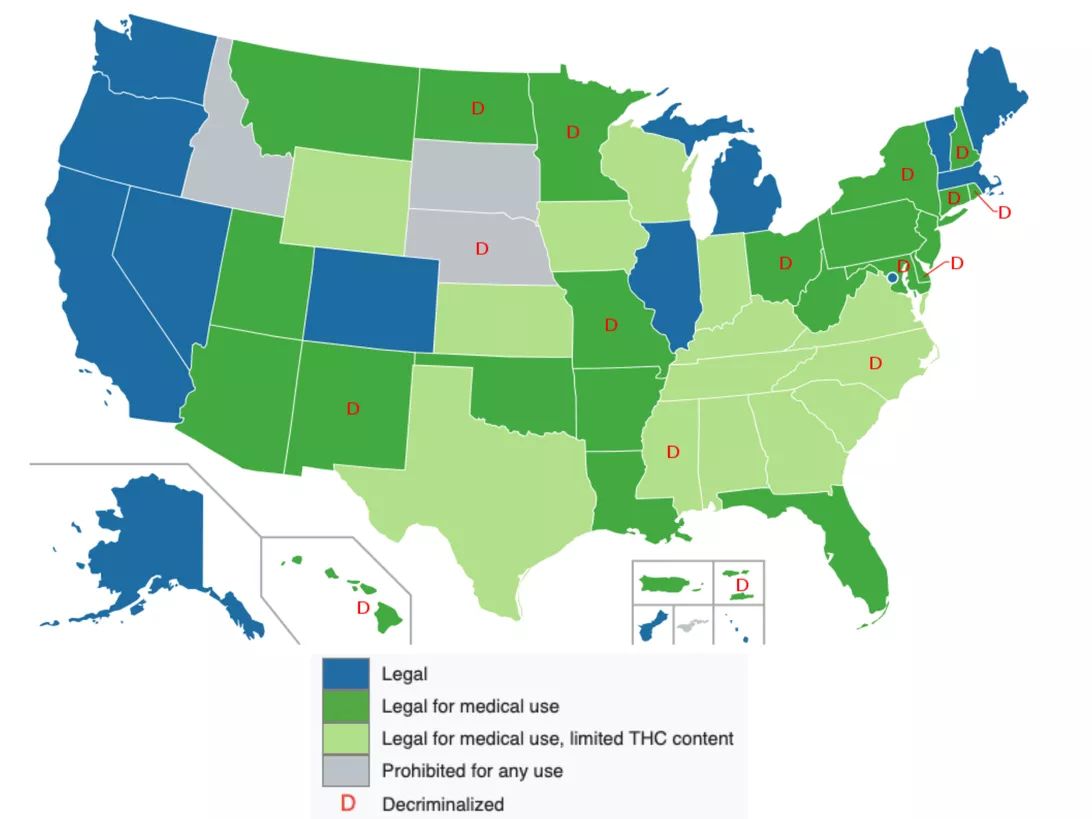
Lokal_Profil/Creative Commons license
Finding approved CBD products
With federal and local cannabis regulations still being worked out, finding CBD products that have the lowest legal risk requires a bit of leg work. Experts recommend being diligent about checking product labels and manufacturers’ websites for any product to understand.
Here’s what to look for:
- Origin of hemp: Look for products made from hemp grown in the United States. Hemp grown overseas puts are at the mercy of that country’s production rules and regulations — or lack there of.
- Certificate of analysis: This document, also called a COA, shows the results of independent lab testing that checks for things like potency (see below) and contaminants. The batch number on the COA should match the number on the product’s label or packaging. Some states make it easy to find this information. For instance, Indiana requires that all CBD products include a QR code on their label that allows users to download the COA to their phone.
- Testing methods: When reading the COA, make sure the lab doing the testing meets “ISO 17025” standards. The testing methods should also have been validated by one of three national regulatory organizations: the Association of Official Agricultural Chemists, the American Herbal Pharmacopoeia, or the U.S. Pharmacopeia.
- Potency: The COA should also confirm that the product contains the amount of CBD and THC listed on its label — both in total and by dose.
If a manufacturer doesn’t have this information or isn’t willing to share it, avoid their products. Not only could you be technically breaking the law by buying it, but research also shows that it’s common for CBD products to be mislabeled, meaning they could be getting more THC than you bargained for or too little CBD to make the product effective.
The information contained in this article is for educational and informational purposes only and is not intended as health or medical advice. Always consult a physician or other qualified health provider regarding any questions you may have about a medical condition or health objectives.



